Fight Against Novel Coronavirus
Oujian supports the fight against novel coronavirus (2019-nCo). Demand for masks and medical supplies has been soared in China due to the coronavirus outbreak. We help the customs clearance of medical devices: face mask, N95 mask, surgical mask, protective garment, protective suit, safety goggles, medical gloves, disinfectant, sanitizer. Epidemic prevention supplies are continuously exported to China. Oujian provides customs clearance services of donations for free.

(Oujian Group providing customs clearance & logistics services in Shanghai for donations to China)
Guidance on Import and Customs Clearance of Anti - Novel Corona-virus Materials
1. Duty Free Certificate for Donations
Step 1: Confirm the person distributing & using the donated goods with the medical institution
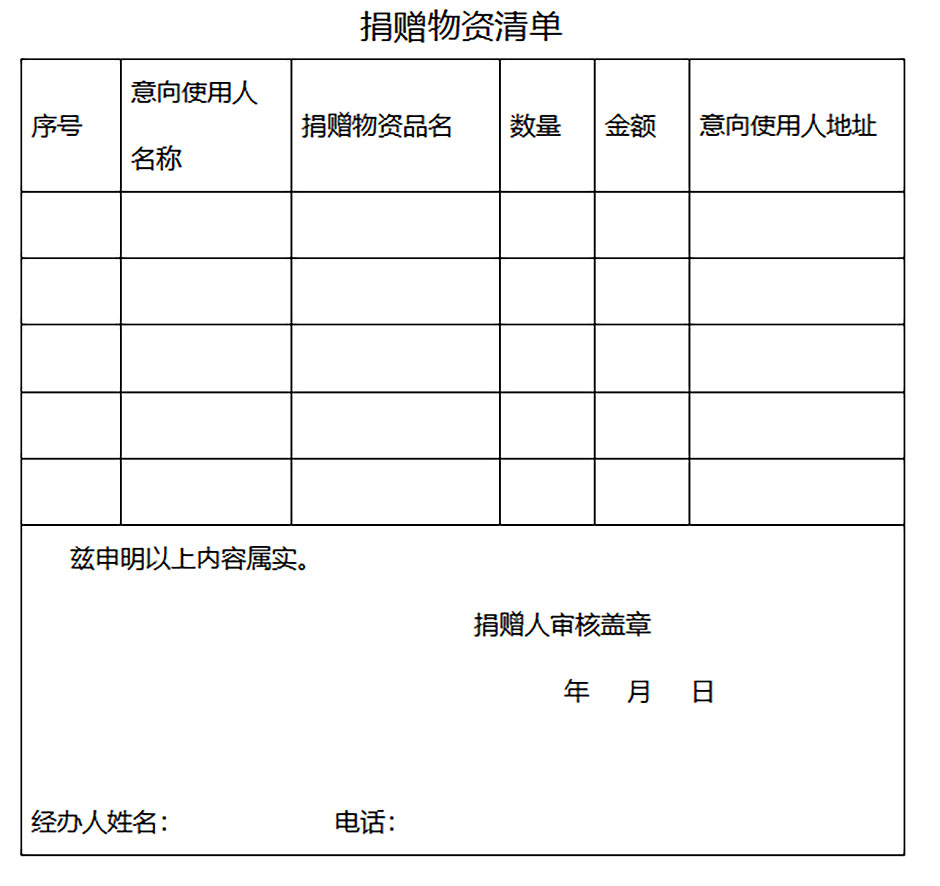
Step 2: Fill out Donation Form (See Annex 1)
1) Letter of Intent for Overseas Donations + List of Donated Goods
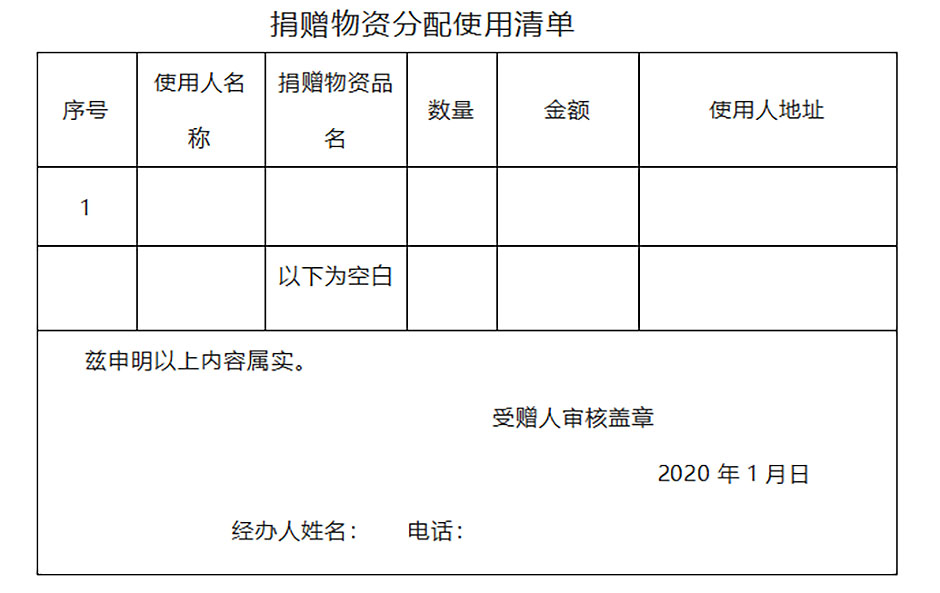
2)Certificate of Recipient Accepting Overseas Donations + Using List of Donated Goods
Step 3: Confirm the donation with a 5A-level social organization or foundation and send them the form for stamp (Color scanned form with stamp of the donee may be temporarily accepted as the declaration form by the customs as a certificate of duty exemption)
5A-level organization in Hubei Province
1) Hubei Charity Federation
Address: 3F, Chumin Building, No. 115 Shouyi Road, Wuchang District, Wuhan, Hubei Province
Contact: Lishuang Zheng +86 135 459 09162
2) Hubei Red Cross Foundation
Address: Room 1101, 11F, ChuShang Building, No.101 Zhongbei Road, Wuchang District, Wuhan, Hubei Province
Contact:+86 181 4069 9039、+86 180 8607 1156、+86 188 7220 3387
3) Hubei Youth Development Foundation
Address: No.5, Dongsan Road, Shuiguo Lake, Wuchang District, Wuhan, Hubei Province
4) Customs Administration in City Wuhai, Hubei Province (Contact: Li Wen +86 156 71666500);
Market Supervision and Administration, Hubei Province (Contact: Yang Li +86 139 08652648);
Drug Administration, Hubei Province (Contact: Yin Xianguo +86 135 07154799
5) Hubei province has issued an announcement, in which 48 organizations have been recognized as qualified recepient of the donations for the novel coronavirus:



Contact:+86 027-8723 3550; 8736 7273
Step 4: Contact Oujian Group for international freight forwarding
Hotline:+86 400 920 1505
Step 5: Contact Oujian Group for customs clearance
Hotline:+86 400 920 1505
(customs clearance consultancy and customs declaration of donations for free)
Step 6: Contact Oujian Group for domestic logistics and distribution
Hotline:+86 400 920 1505
(Inland logistics and distribution of donations to Wuhan for free)
2. Customs Declaration of Donations/General Trade/Other Import and Export
1) Documents:Waybill, Agency Agreement (required in case of entrustment of agent)、invoice、packing list(See Annex 2)
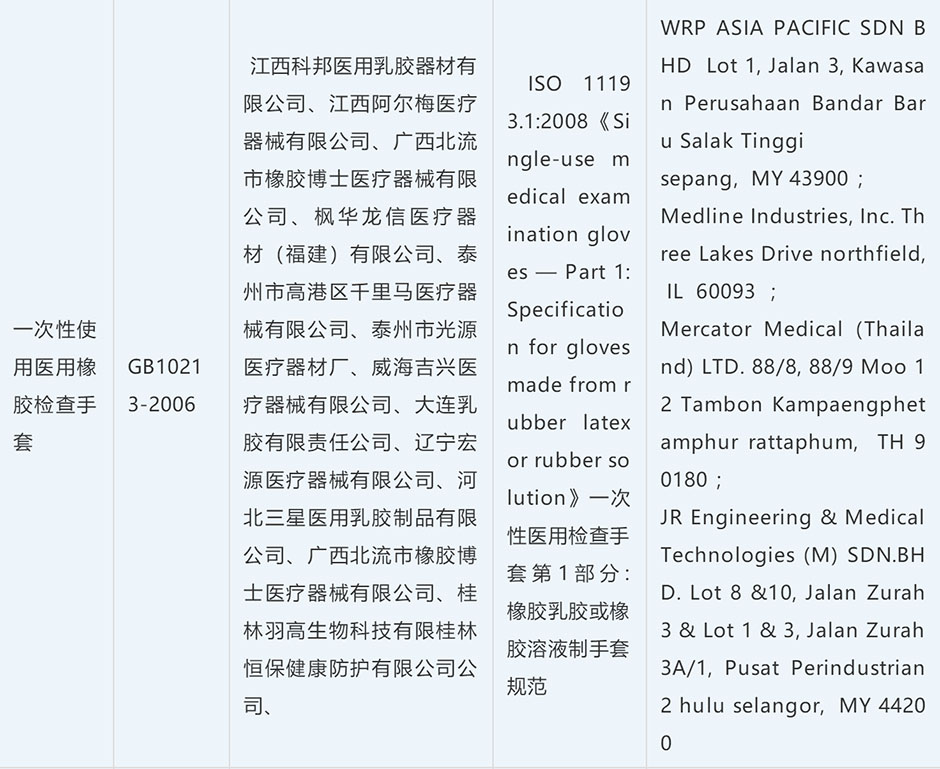
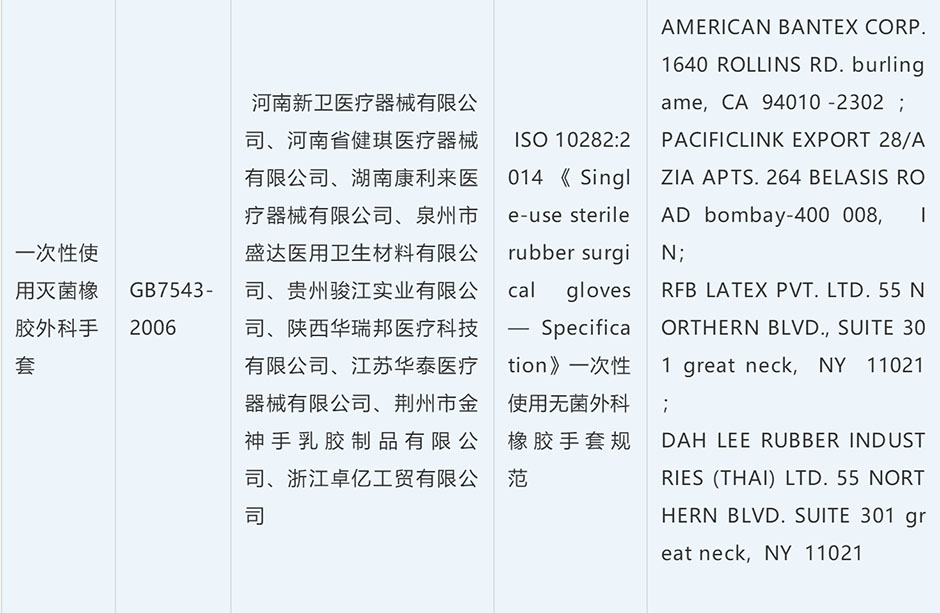
Most needed donations/ medical supplies / epidemic prevention supplies: mask, face mask, surgical mask, N95 Mask, protective garment, protective suit, goggle, safety goggle, glove, medical glove, disinfectant, sanitizer.



2) Declaration Elements & Duties
| Name | HS Code | Declaration Element | MFNT | VAT | Imposed Tariff on Products from the US |
| Mask | 63079000 | Brand, ingredient | 6% | 13% | 25% |
| Goggle | 90049090 | Brand, purpose, type(discoloration, etc.), model | 7% | 13% | 25% |
| Protective Suit | 62101030 | Weaving method [weaving, etc.), category [jacket, trousers, shorts, etc.], category [male, female], Ingredients, brand name [both in Chinese & foreign language] | 8% | 13% | 25% |
| Vulcanized Rubber Glove | 40151900 | purpose, brand, ingredients[gum content], specification [S, M, XL, etc.] | 10% | 13% | 25% |
3) Medical Device Certification
Before import, the supervision category of imported goods could be inquired from the NMPA website according to the "Measures for the Administration of Inspection and Supervision of the Imported Medical Instruments"
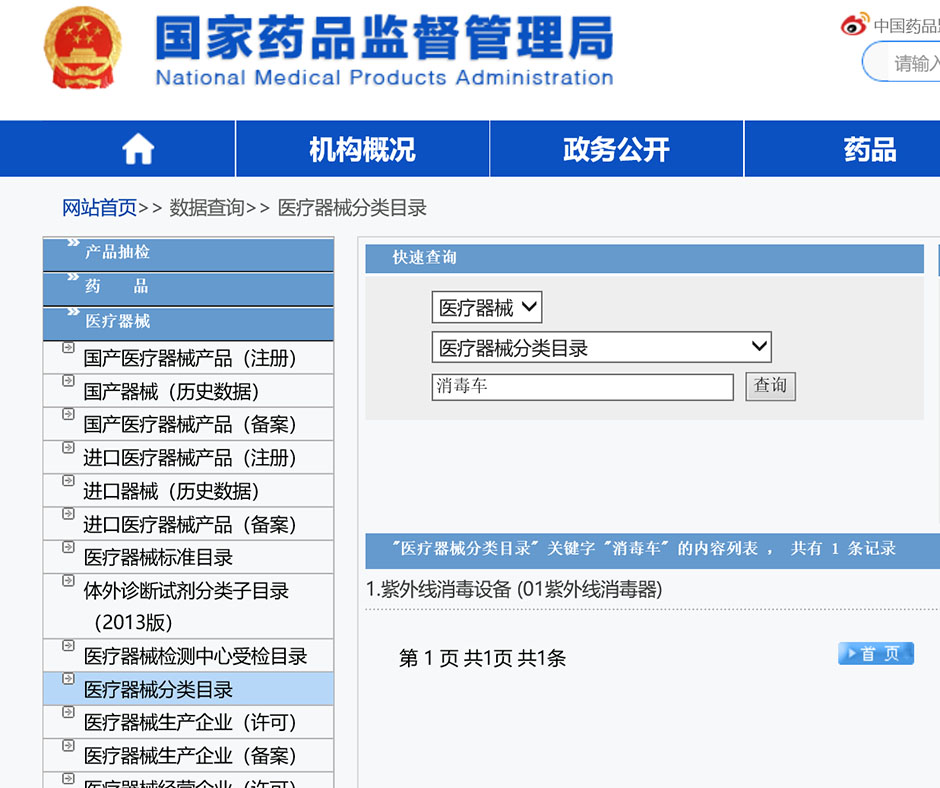

Category I equals low risk level. “Category I Medical Device Registration Certificate" need to be submitted by customs declaration

Category II equals medium risk level. "Medical Device Registration Certificate of PRC" need to be submitted by customs declaration. Meanwhile the business operator need to register at the local food and drug supervision and administration department.


Category III equals high risk level. "Medical Device Registration Certificate of PRC" need to be submitted by customs declaration. Meanwhile the business operator need to apply for medical device business certificate at the local food and drug supervision and administration department.



4) Customs Clearance without Medical Device Certificate
A. Self-used Goods with Taxation: enterprises importing anti-epidemic materials for self-use shall provide regular waybill, invoice, packing list and an additional letter of commitment, that the imported goods are for non-medical use. Before release customs may carry out inspection of the goods to check the name, composition, model and whether it’s medical-use or not.
B. Donated materials: According to No. 17 announcement of GACC 2020 , each directly under the customs-related customs clearance site set up imported donated materials rapid customs clearance special acceptance window and green channel, the implementation of rapid inspection and release. In case of emergency, you can register for release, and then in accordance with the provisions to complete the relevant procedures. That is to say, for special items and medical devices that have not been registered in our country, they may apply to the competent departments such as the Drug Enforcement Administration for relevant supporting documents and apply to the Customs for release. If it is a special case, you may apply for release first, and then the recipient and the user to make up the relevant supporting documents.
In practice, the recipient may issue a letter of commitment from the charity, and commit to go to the Drug Administration for quality testing and testing after passing the requirements of the Drug Enforcement Administration before use. After the customs release, the grantee shall, with the certificate of listing of overseas medical devices, the overseas inspection report, the commitment to product quality and safety, the description of the product in accordance with local standards or substantial equivalent to the domestic product, the product label and the specification (all of which are provided by the above documents Chinese) to the Drug Enforcement Administration for the relevant certificate, If the test results do not meet the medical standards may not be used for medical use.
Contact Person for Certification Process of City Drug Administration:
Lijun Hu +86 189 1760 2139
C. Taxation for Sale: Foreign trade enterprises imported in large quantities for sale, the import enterprise must have a medical certificate in line with various categories, including medical device registration certificate, medical device record certificate, medical device business license, etc., the customs can not refer to the donated materials for the record registration and release.
D. Personal items mailed import method: in the name of personal mail medical supplies (including masks, goggles, etc.) not more than 1000 yuan, overseas purchase can be directly sent to relatives and friends in China, the recipient correctly handwritten name of the Chinese Pinyin; The recipient's name or company's pinyin spelling or English name must be correctly written;
Remark: Personal items are not recommended to use too large cartons, the maximum weight is recommended not more than 30 kg;
Foreign Medical Registration Certificate:

Foreign inspection reports:

Letter of Commitment:

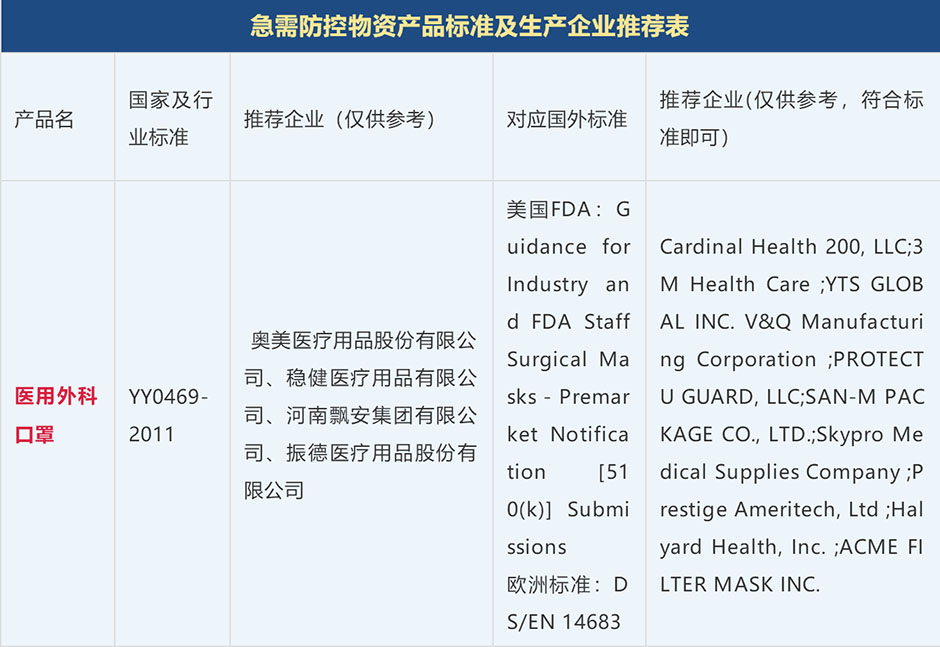
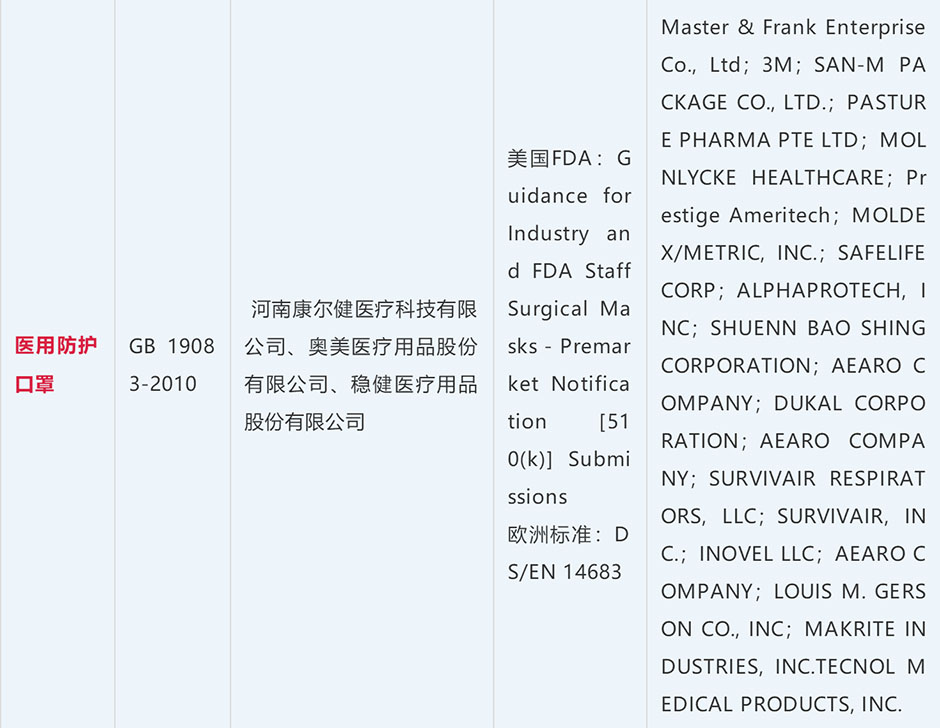

Chinese Label Requirements:

Annex 1:
Letter of intent for overseas material donation

List of detailed donations
Certificate of acceptance of overseas donations for novel coronavirus by recipients
List of use for the distribution of donated materials
Annex 2: Sample of Commercial Invoice & Packing List
| COMMERCIAL INVOICE/PACKING LIST | |||||||
| FORM: | TO: | ||||||
| CIF WUHAN | |||||||
| 联系人: | |||||||
| PACKING | TRANSPORTATION | ||||||
| CARTON/PALLET/WOODEN CASE | BY AIR | ||||||
| FINAL DESTINATION | TED | ||||||
| WUHAN | 25-Jan-20 | ||||||
| DESCRIPTION | QTY数量 | N.W净重 | G.W毛重 | AMOUNT金额 | |||
| Mask | 0 | 0 | 0 | ||||
| Protective clothing | 0 | 0 | 0 | ||||
| Goggles | 0 | 0 | 0 | ||||
| Total | 0 | 0 | 0 | ||||
| SELLER: | |||||||
| 件数: | |||||||









